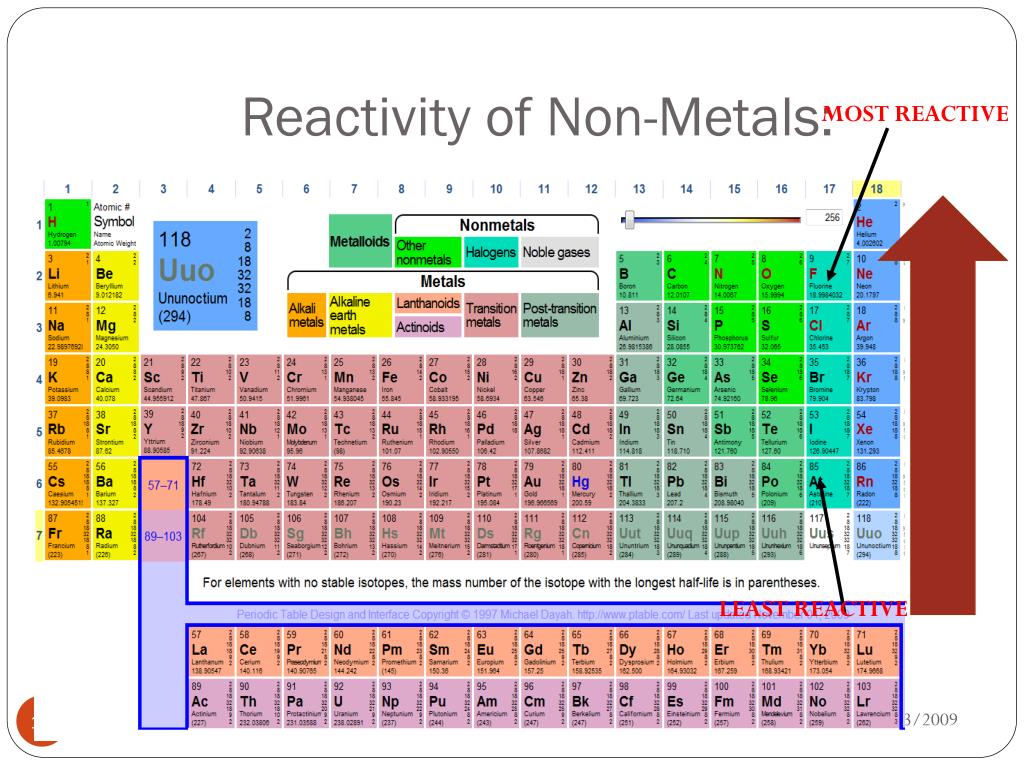
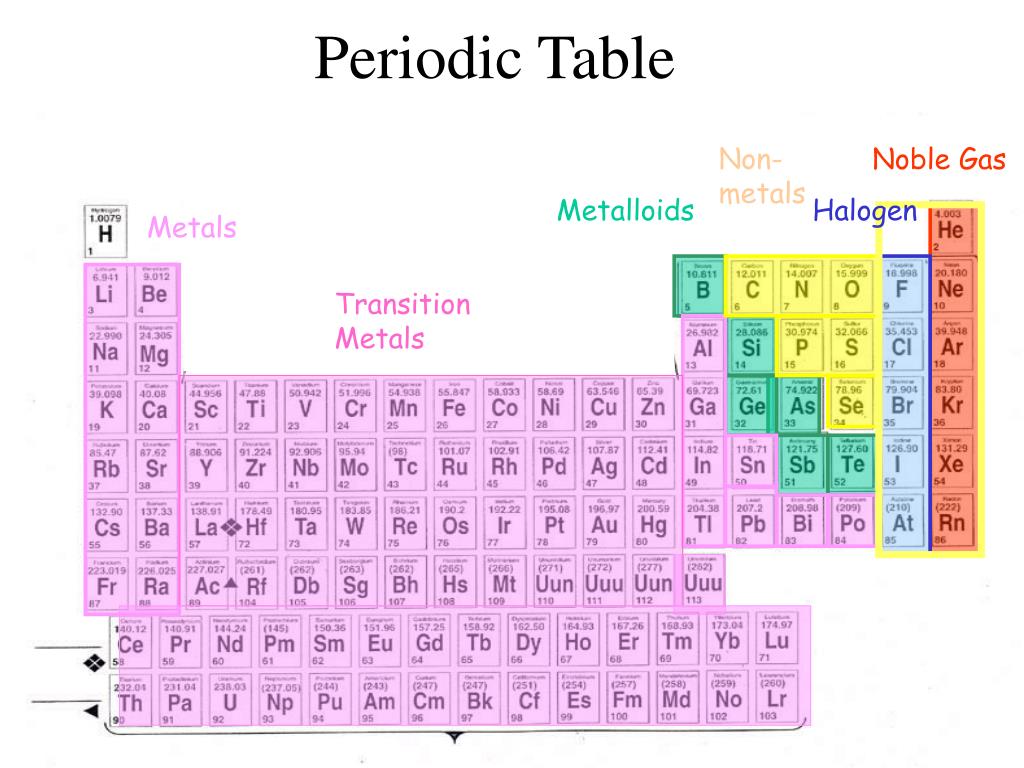
 So it has highest electronegativity (it has maximum tendency to attract the electrons pair)Īnd because of this, fluorine has a highest tendency to react with any other element to form a compound.Įven fluorine reacts with noble gas like xenon, and forms compounds like XeF4 (Xenon tetrafluoride) and XeF6 (Xenon hexafluoride) In other words, Fluorine is at the top of the halogen group and it has less atomic size plus it needs only one electron to complete the octet. The short answer: Fluorine is a highly reactive nonmetal on the entire Periodic table.Īctually the halogens are the most reactive nonmetals, but we know that as we move down the group, the electronegativity decreases. Most reactive nonmetal on the Periodic table Keep reading… more important topics are on the way. Atomic number Symbol Name of element 1 H Hydrogen 2 He Helium 6 C Carbon 7 N Nitrogen 8 O Oxygen 9 F Fluorine 10 Ne Neon 15 P Phosphorus 16 S Sulfur 17 Cl Chlorine 18 Ar Argon 34 Se Selenium 35 Br Bromine 36 Kr Krypton 53 I Iodine 54 Xe Xenon 85 At Astatine 86 Rn Radon Here is a complete list of all the 18 nonmetals on the Periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
